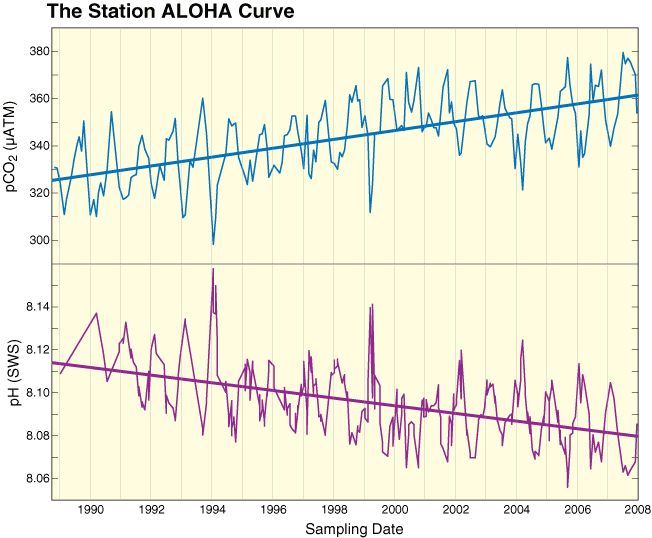
One of the least-understood consequences of increasing carbon dioxide concentrations in the atmosphere is that the oceans are becoming more acidic. This is because CO2 in the air dissolves in seawater to form carbonic acid � a weak acid that makes the oceans slightly more acidic. The rate of change is extremely rapid and it is expected that by the end of this century, the oceans will be more acidic than they have been for more than 20 million years. Over geological time scales, the pH of the seas has changed significantly in response to variations in atmospheric CO2. Indeed, the ocean has in the past been more acidic than we expect it to become over the coming decades. But what is different this time is the speed of change. Ecosystems have proved their ability to accommodate change when it is gradual, usually over hundreds of thousands of years. We do not know how well the marine ecosystem will adapt to changes that will occur over decades.
Microbes are the most important organisms in the sea. In contrast to the land, where plants are large and long-lived, in the seas most of the primary production comes from microscopic algae, or phytoplankton. These have tiny biomass and their generations last only days. The productivity of this phytoplankton depends in turn on bacteria and archaea to regenerate nutrients. So it was a priority to determine how marine microbes would respond to a high-CO2 world.
Microbes control biogeochemical cycles and keep the planet habitable. This means we need to know how microbial populations will respond to rapid climate change. We need to do experiments to understand how marine microbes will respond to pH change.
Such experiments will be complex because pH varies naturally over the course of a year. When phytoplankton populations begin to grow in the spring they consume CO2 that is dissolved in seawater. The result is that pH increases for a period of a few weeks. The dissolved CO2 gradually increases as a result of bacterial activity and the pH declines to a more typical values. In a high CO2 world, this seasonal variability will still exist but the microbial populations will be exposed to a different range of pH values than in the present day.
EDIT
http://cmore.soest.hawaii.edu/oceanacidification/index.htm