Welcome to DU!
The truly grassroots left-of-center political community where regular people, not algorithms, drive the discussions and set the standards.
Join the community:
Create a free account
Support DU (and get rid of ads!):
Become a Star Member
Latest Breaking News
General Discussion
The DU Lounge
All Forums
Issue Forums
Culture Forums
Alliance Forums
Region Forums
Support Forums
Help & Search
Health
Related: About this forumMeningitis: drug firm escaped tough sanctions before outbreak, files show
http://www.guardian.co.uk/society/2012/oct/23/meningitis-firm-escaped-sanctions-outbreak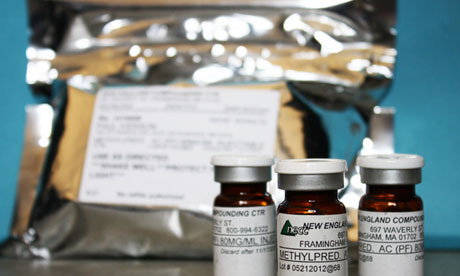
Regulators stopped short of imposing the harshest sanctions on the NECC even as the list of complaints continued to grow, the documents show. Photograph: AP
The company linked to the deadly meningitis outbreak had repeatedly escaped harsh sanctions from health regulators in the years leading up to the scandal, raising fresh questions about oversight of the compounding pharmacy sector.
Problems at the New England Compounding Center in Framingham, Massachusetts stretched as far back as 1999, according to state records released amid a wave of legal action from patients who received contaminated or potentially contaminated steroid injections made by NECC.
One lawyer told the Guardian that information in the documents could expand the scope of the lawsuits against the company to include punitive damages.
On Tuesday the US Centers for Disease Control updated the number of cases to 304, including one new case in Georgia, bringing the number of states affected to 17. No new deaths were reported.
InfoView thread info, including edit history
TrashPut this thread in your Trash Can (My DU » Trash Can)
BookmarkAdd this thread to your Bookmarks (My DU » Bookmarks)
1 replies, 925 views
ShareGet links to this post and/or share on social media
AlertAlert this post for a rule violation
PowersThere are no powers you can use on this post
EditCannot edit other people's posts
ReplyReply to this post
EditCannot edit other people's posts
Rec (1)
ReplyReply to this post
1 replies
 = new reply since forum marked as read
Highlight:
NoneDon't highlight anything
5 newestHighlight 5 most recent replies
= new reply since forum marked as read
Highlight:
NoneDon't highlight anything
5 newestHighlight 5 most recent replies
Meningitis: drug firm escaped tough sanctions before outbreak, files show (Original Post)
xchrom
Oct 2012
OP
xchrom
(108,903 posts)1. Pharmacy in Meningitis Deaths Found Mold in January
http://www.bloomberg.com/news/2012-10-26/pharmacy-in-meningitis-outbreak-found-with-mold-bacteria.html
New England Compounding Pharmacy Inc., the company linked to a U.S. meningitis outbreak, discovered elevated levels of mold and bacteria at its drug- making facility in January and didn’t act, regulators said.
When the contamination was found to have exceeded limits the pharmacy itself set for mold and bacteria, “no documented corrective actions were taken,” the Food and Drug Administration said in a preliminary report that cites company documents. The FDA’s investigators uncovered tainted steroids and unsanitary conditions during inspections that began Oct. 1 at the facility in Framingham, Massachusetts, the agency said.
“It wouldn’t be proper for us to characterize this firm beyond the factual observations at this time,” Howard Sklamberg, the FDA’s deputy associate commissioner for regulatory affairs, said yesterday on a call with reporters. “This is part of an investigation. A broader investigation is where an agency characterizes and draws conclusions.”
The pharmacy suspended operations Oct. 3 after state and federal regulators began to link one of its steroid products, methylprednisolone acetate, to fungal meningitis infections that have now killed 25 people. The company, which used the name New England Compounding Center, recalled more than 17,000 doses of the steroid sold to at least 75 hospitals and clinics in 23 states. The government has said about 14,000 people received shots of the steroid, which is injected into the spinal cavity to relieve neck and back pain.
New England Compounding Pharmacy Inc., the company linked to a U.S. meningitis outbreak, discovered elevated levels of mold and bacteria at its drug- making facility in January and didn’t act, regulators said.
When the contamination was found to have exceeded limits the pharmacy itself set for mold and bacteria, “no documented corrective actions were taken,” the Food and Drug Administration said in a preliminary report that cites company documents. The FDA’s investigators uncovered tainted steroids and unsanitary conditions during inspections that began Oct. 1 at the facility in Framingham, Massachusetts, the agency said.
“It wouldn’t be proper for us to characterize this firm beyond the factual observations at this time,” Howard Sklamberg, the FDA’s deputy associate commissioner for regulatory affairs, said yesterday on a call with reporters. “This is part of an investigation. A broader investigation is where an agency characterizes and draws conclusions.”
The pharmacy suspended operations Oct. 3 after state and federal regulators began to link one of its steroid products, methylprednisolone acetate, to fungal meningitis infections that have now killed 25 people. The company, which used the name New England Compounding Center, recalled more than 17,000 doses of the steroid sold to at least 75 hospitals and clinics in 23 states. The government has said about 14,000 people received shots of the steroid, which is injected into the spinal cavity to relieve neck and back pain.